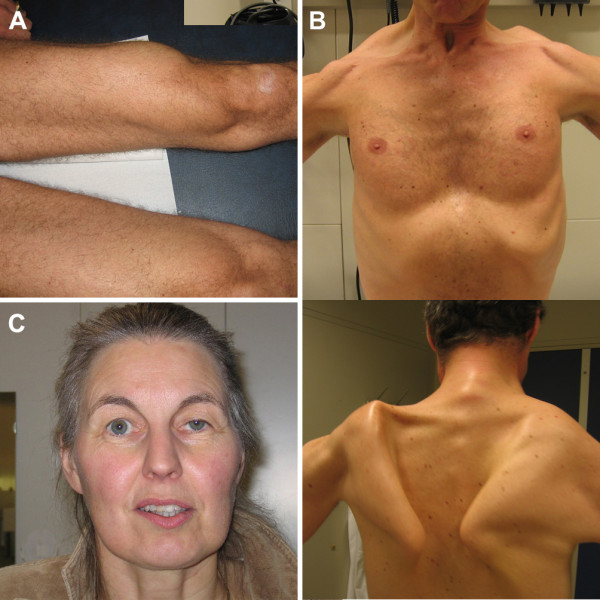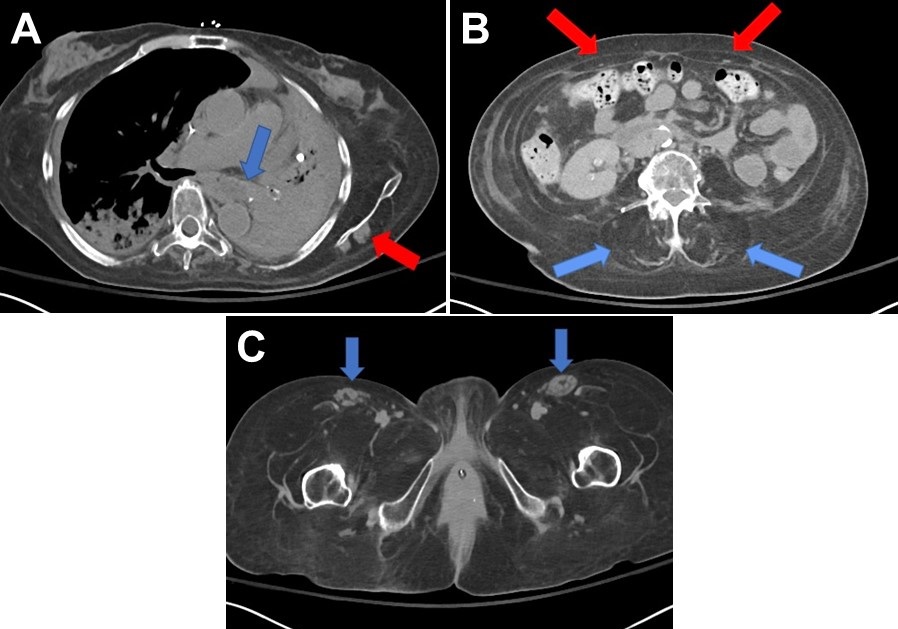
Southwest Journal of Pulmonary, Critical Care and Sleep - Imaging - Medical Image of the Month: Late-Onset Pompe Disease

Late-onset Pompe disease (LOPD): Correlations between respiratory muscles CT and MRI features and pulmonary function - ScienceDirect
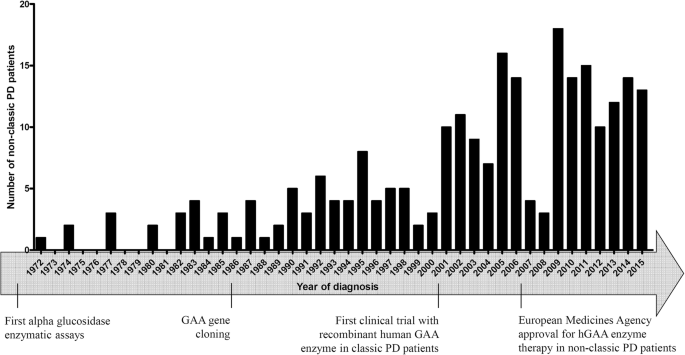
Late-onset Pompe disease in France: molecular features and epidemiology from a nationwide study | Journal of Inherited Metabolic Disease
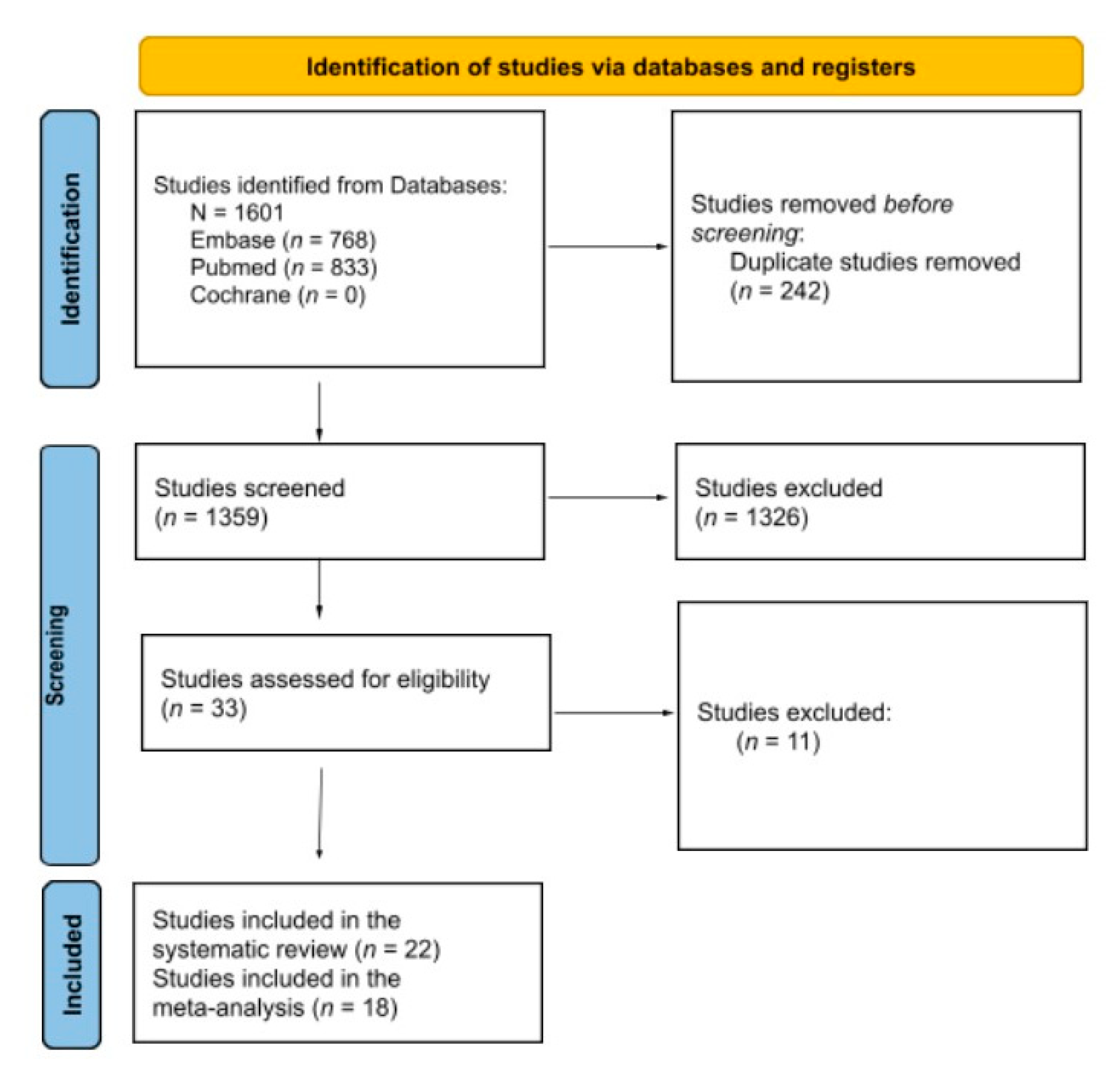
JCM | Free Full-Text | A Systematic Review and Meta-Analysis of Enzyme Replacement Therapy in Late-Onset Pompe Disease

Investigating Late-Onset Pompe Prevalence in Neuromuscular Medicine Academic Practices | Neurology Genetics

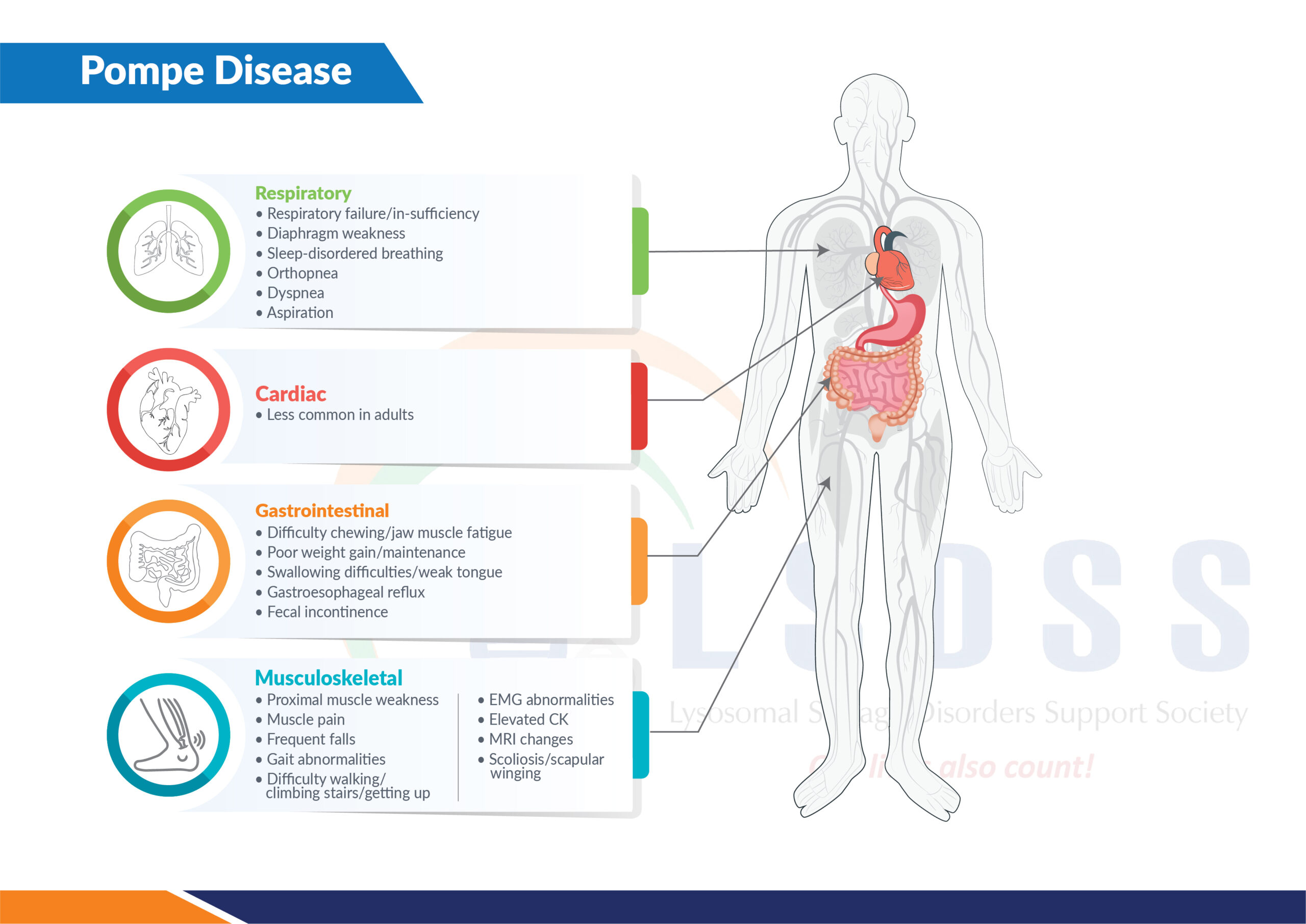
Blunted Hypercapnic Respiratory Drive Response in Subjects With Late-Onset Pompe Disease | Respiratory Care

Pathophysiology of late-onset Pompe disease. Abbreviations: GAA, acid... | Download Scientific Diagram

Safety and efficacy of avalglucosidase alfa versus alglucosidase alfa in patients with late-onset Pompe disease (COMET): a phase 3, randomised, multicentre trial - The Lancet Neurology

Marrow on X: "The @US_FDA Approved Nexviazyme (avalglucosidase alfa-ngpt) For Late-Onset Pompe Disease Recommended dose: 20 mg/kg for patients ≥30 kg OR 40 mg/kg for patients <30 kg | #medtwitter https://t.co/CZN4NK0X2n" /

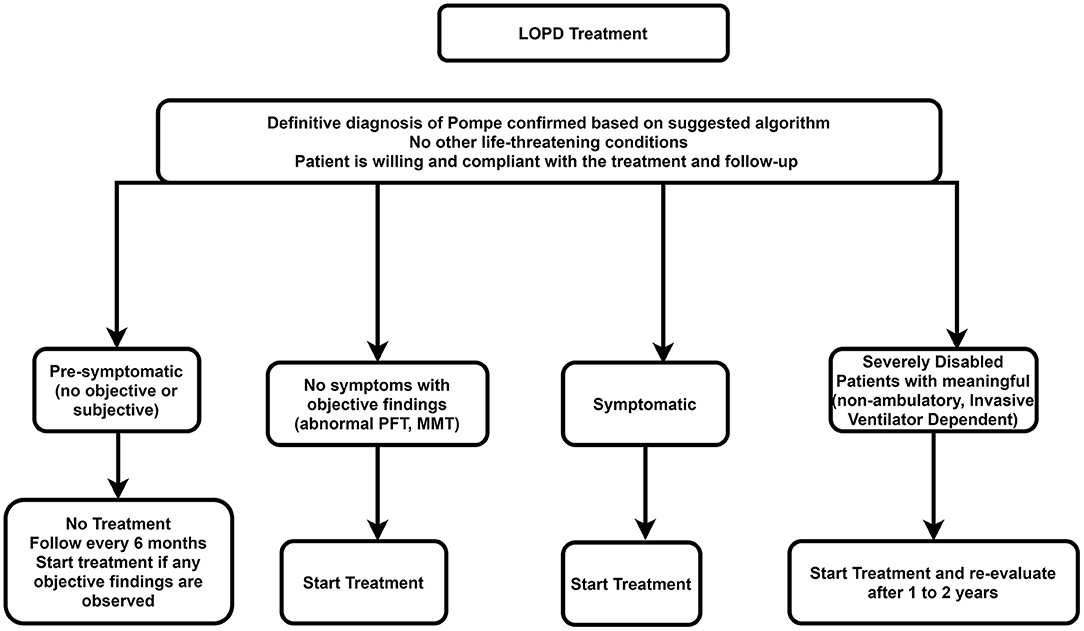
Managing Late-onset Pompe Disease in the Era of Newborn Screening: Considerations for Disease Monitoring and ERT Initiation | Catalyst Medical Education

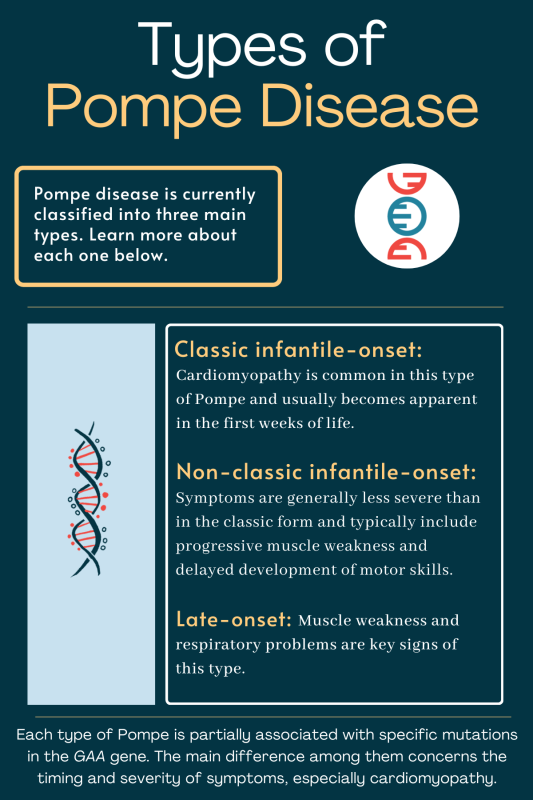
Childhood Pompe disease: clinical spectrum and genotype in 31 patients | Orphanet Journal of Rare Diseases | Full Text

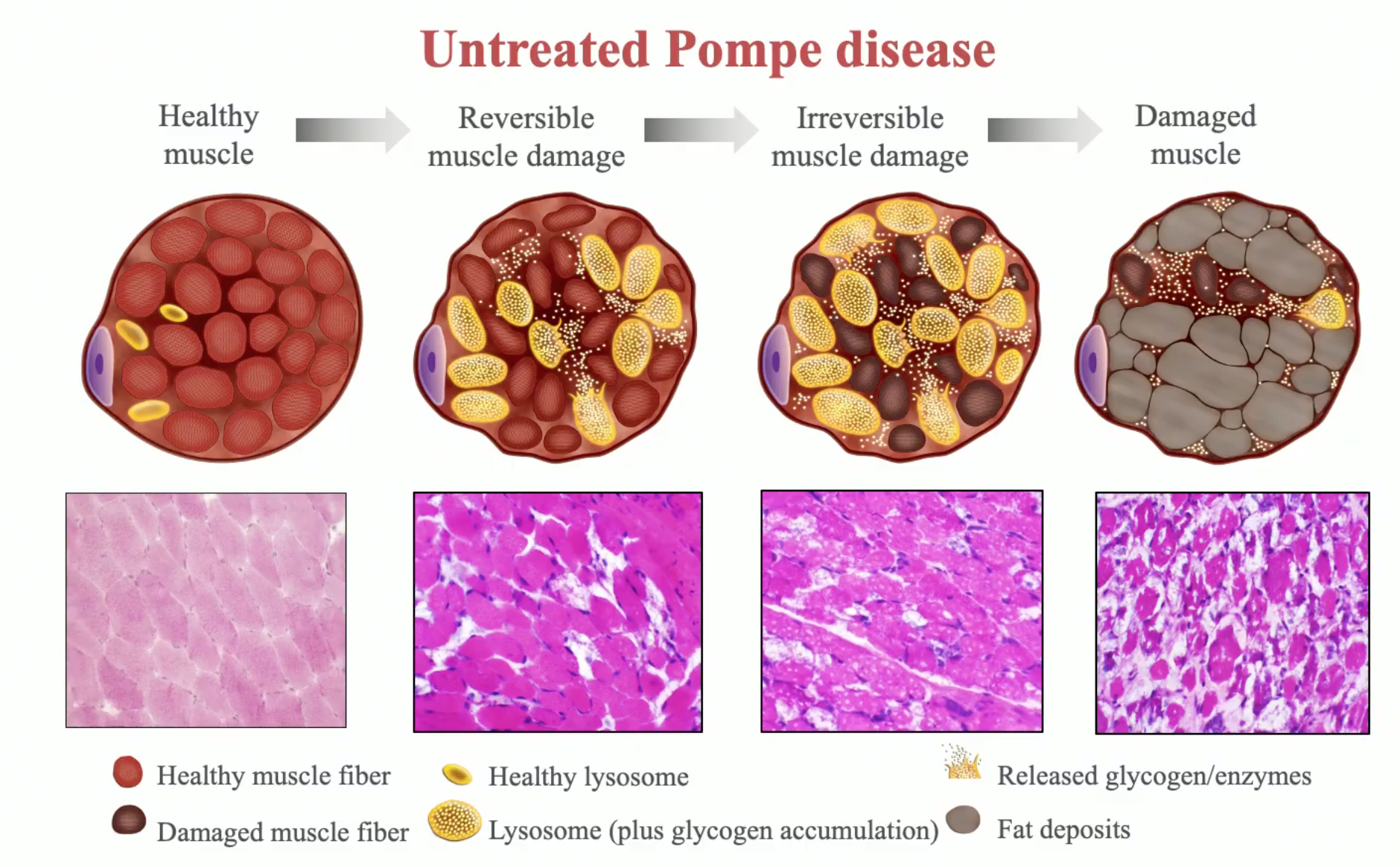
Follow‐up of late‐onset Pompe disease patients with muscle magnetic resonance imaging reveals increase in fat replacement in skeletal muscles - Nuñez‐Peralta - 2020 - Journal of Cachexia, Sarcopenia and Muscle - Wiley