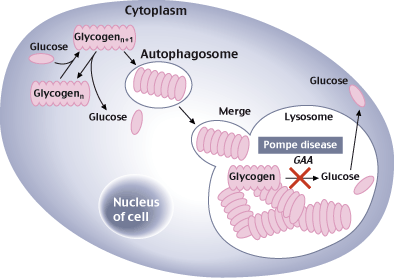
Malattia di Pompe, via libera europeo all'uso di avalglucosidasi alfa come nuovo potenziale standard di cura - AboutPharma

Sanofi invests US$150m to acquire oral Pompe disease drug candidate from Maze Therapeutics | HealthCare Middle East & Africa Magazine

Sanofi termina l'accordo sulla licenza del farmaco dopo le obiezioni della FTC statunitense -11 dicembre 2023 alle 22:33 | MarketScreener
FDA aprueba Nexviazyme® de Sanofi Genzyme, una nueva e importante opción de tratamiento para la enfermedad de Pompe